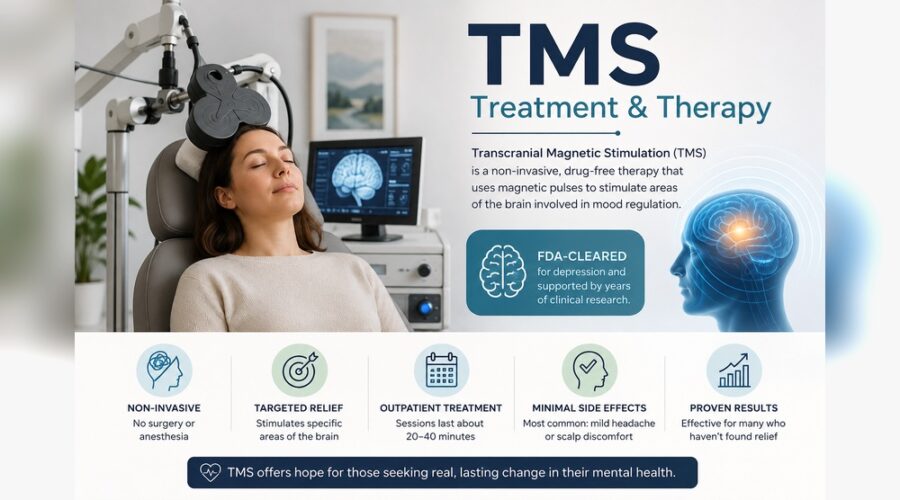
TMS Therapy: A Complete 2026 Guide to Treatment, Cost, Insurance & Devices
TMS therapy is the rare interventional psychiatry treatment where insurance is the default — not the exception. Most major commercial plans, Medicare Part B, and a growing list of state Medicaid programs cover it for treatment-resistant depression. Patients drive themselves to and from sessions, work the same day, and finish a course in 6 to 9 weeks without anesthesia, IV access, or controlled substances.
This guide explains how TMS therapy works in 2026: the FDA-cleared conditions it treats, the device and protocol differences that matter, what insurance actually covers (and what it denies), realistic costs, side effects, the patient-experience advantages over ketamine, and the decision framework for choosing it. Reviewed by Angelica Bottaro, medical writer and verifier.
Key Takeaways
| FDA status | FDA-cleared (510(k) and De Novo pathways) since 2008 for treatment-resistant depression. Not “FDA-approved” — TMS devices are cleared, not PMA-approved. |
| Insurance coverage | Covered by most BCBS, Aetna, Cigna, UnitedHealthcare, Anthem, Humana, Tricare, and Medicare Part B plans for major depressive disorder. Pre-authorization required, usually after 2 to 4 failed antidepressant trials. |
| Course length | 36 sessions over 6 to 9 weeks (standard rTMS). 19 to 37 minutes per session. Newer iTBS protocols compress sessions to 3 minutes. Accelerated SAINT protocol delivers 50 sessions in 5 days. |
| Cost | Cash-pay: $300 to $500 per session, $7,200 to $15,000 per course. With insurance: $0 to $250 per session, $500 to $3,000 typical out-of-pocket for a full course. |
| Patient experience | No anesthesia. No sedation. No IV. No driver required. No dissociation. Patients return to work the same day. No controlled-substance handling. |
| Response rate | 50 to 60 percent for treatment-resistant depression. Remission rate 30 to 40 percent. Two-thirds of responders maintain benefit at 6 to 12 months. |
What TMS Therapy Is — And What It Isn’t
Transcranial magnetic stimulation uses targeted magnetic pulses to stimulate nerve cells in regions of the brain involved in mood regulation. A coil is placed against the scalp over the dorsolateral prefrontal cortex, and brief magnetic fields induce small electrical currents in the underlying cortex. The pulses are non-invasive and outpatient. There is no anesthesia, no sedation, and no recovery period. Patients sit in a chair, fully awake, for the duration of each session.
TMS is sometimes confused with electroconvulsive therapy (ECT). The two are not similar. ECT requires general anesthesia, induces a controlled seizure, and is delivered in a hospital. TMS produces no seizure (the rate of accidental seizures is roughly 1 in 89,000 sessions per current real-world data) and does not require any anesthesia or memory-affecting interventions. Patients who tolerate ECT poorly often tolerate TMS easily. Read more in our comparison of TMS and ECT for treatment-resistant depression.
TMS also differs sharply from ketamine therapy. Ketamine produces a dissociative experience requiring 1 to 2 hours of monitored recovery and a chaperone for transit home. TMS produces no perceptual changes; patients drive themselves and return to work the same day. The two treatments serve overlapping but distinct populations — see our breakdown of TMS vs ketamine for depression.
FDA-Cleared Conditions and the Adolescent Update
The FDA has cleared TMS devices for an expanding list of psychiatric conditions since the original 2008 indication for treatment-resistant depression. The 2024 and 2025 adolescent clearances represent the most consequential recent developments — TMS is now an FDA-cleared option for ages 15 to 21, not 12 and up.
| Year | Indication | Device |
|---|---|---|
| 2008 | Adult major depressive disorder | NeuroStar (first); subsequently most major manufacturers |
| 2018 | Obsessive-compulsive disorder (adjunct, adults) | BrainsWay Deep TMS H7 — only standalone OCD clearance via De Novo pathway |
| 2018 | iTBS (3-minute protocol) for MDD | MagVenture, then most manufacturers |
| 2020 | Short-term smoking cessation in adults | BrainsWay Deep TMS H4 |
| 2021–2022 | Anxious depression (comorbid anxiety in MDD) | BrainsWay (2021), NeuroStar (2022) |
| 2022 | Accelerated MRI-guided iTBS for TRD | Magnus Medical SAINT system |
| 2024 | Adolescent MDD adjunct, ages 15 to 21 | NeuroStar (March 2024) — first TMS clearance for adolescents |
| 2025 | Adolescent MDD adjunct, ages 15 to 21 | BrainsWay Deep TMS (November 2025) |
| 2025 | Combined MDD, OCD, and anxious depression | Magstim Horizon Inspire |
For obsessive-compulsive disorder, only BrainsWay Deep TMS using the H7 coil holds a standalone FDA clearance. MagVenture’s Cool D-B80 has an OCD adjunct clearance, but Deep TMS H7 remains the only TMS device cleared specifically for OCD via the De Novo pathway. If you are seeking TMS for OCD, ask the clinic which device they use — this matters for both insurance coverage and treatment fit.
Devices and Protocols — What Patients Should Know
Five major device manufacturers serve the U.S. TMS market, and each uses different coil designs and clears different protocols. Patients searching for accelerated TMS, OCD treatment, or pediatric care should ask which device a clinic operates before booking — the answer determines what conditions can be treated on-label.
| Device | Coil Type | Notable Clearances |
|---|---|---|
| NeuroStar (Neuronetics) | Figure-8, focal | MDD adults (2008), anxious depression (2022), adolescent MDD 15–21 (2024). Market leader, roughly 60 percent U.S. share. |
| BrainsWay Deep TMS | H-coil family (H1, H4, H7) — non-focal, deeper penetration | MDD (2013), OCD (2018, only standalone TMS-for-OCD clearance), smoking cessation (2020), anxious depression (2021), adolescent MDD 15–21 (2025). |
| MagVenture | Figure-8 and butterfly variants (Cool-B65, Cool-DB-80) | MDD (multiple session lengths including 3-min iTBS, 19-min, 37-min). Markets the 3-min protocol as “Express TMS.” |
| Magstim Horizon | Figure-8 with StimGuide navigation | MDD, OCD, anxious depression. The 2025 Horizon Inspire combines all three indications. First FDA-cleared TMS navigation system (2019). |
| Nexstim NBT | Navigated with individualized MRI | MDD; specialty in stroke recovery and motor mapping. Smaller U.S. footprint. |
| Magnus Medical SAINT | Uses MagVenture hardware + Magnus targeting software | Accelerated MRI-guided iTBS for TRD (2022). Clinic must be Magnus-credentialed; fewer than 10 U.S. clinics currently are. |
The Protocol Differences That Matter
Standard 10 Hz rTMS delivers 3,000 magnetic pulses over 19 to 37 minutes per session. Intermittent theta burst stimulation (iTBS), FDA-cleared in 2018, delivers 600 pulses in just 3 minutes — non-inferior to standard rTMS in the THREE-D trial and far easier to fit into a workday. “Express TMS” is the MagVenture marketing name for the same iTBS protocol.
Accelerated TMS compresses the standard course into 1 to 5 days by stacking multiple sessions per day. The most rigorously studied accelerated protocol is the Stanford SAINT (also called SNT) regimen — 50 iTBS sessions delivered over 5 consecutive days, with 79 percent remission in the original randomized trial. SAINT requires Magnus credentialing; not every clinic claiming “accelerated TMS” offers it. As of July 2025, CMS reimburses hospitals $19,703 for the full SAINT outpatient protocol, signaling broader payer adoption.
The Treatment Course — What Six Weeks Actually Looks Like
The standard TMS course is 36 sessions over 6 to 9 weeks at five sessions per week. Patients typically taper down in the final two weeks. Each session takes 19 to 37 minutes for standard rTMS or just 3 minutes for iTBS, plus check-in time. The first session adds 20 to 60 minutes for motor threshold mapping.
Step 1: Insurance Pre-Authorization (1 to 3 weeks)
Before treatment begins, the clinic submits a prior-authorization request to your insurer. The request typically requires: a confirmed DSM-5 major depressive disorder diagnosis from a psychiatrist, documented failure of 2 to 4 adequate antidepressant trials (most carriers want at least 6 weeks at therapeutic dose for each), a baseline depression rating scale (PHQ-9, HAM-D, or BDI), and evidence of inadequate response to or contraindication for psychotherapy. Industry data suggests 30 to 50 percent of initial requests are denied. The most successful appeal mechanism is a peer-to-peer review between your clinic’s psychiatrist and the insurer’s medical reviewer. Read the full breakdown of which insurance companies cover TMS therapy.
Step 2: Motor Threshold Mapping (first session)
Motor threshold mapping happens once at the first treatment visit. The clinician delivers single magnetic pulses to the motor cortex and finds the minimum stimulation intensity that elicits a thumb or finger twitch. This motor threshold value sets the dosing for every subsequent session — typically 110 to 120 percent of motor threshold. Re-mapping is non-routine; clinicians repeat it only if response stalls, headaches escalate, or device parameters change.
Step 3: Daily Treatment Sessions
Daily sessions run Monday through Friday. The patient sits in a recliner. Hearing protection is mandatory because the magnetic pulses are loud. The coil is positioned against the scalp; the operator initiates pulses; the patient feels a tapping sensation on the scalp during stimulation. There is no medication, no IV, and no need to fast. Patients commonly read, listen to music, or scroll their phone during the session.
Step 4: Drive Home, Return to Work
This is the largest patient-experience advantage TMS holds over other interventional psychiatry treatments. Patients drive themselves to and from each session. They return to work, school, or their normal day immediately afterward. There is no chaperone requirement, no recovery period, and no integration block. Many patients schedule sessions during a lunch break.
Why TMS — Ten Patient-Experience Advantages
For patients comparing TMS against ketamine therapy, Spravato, or ECT, the differences in daily life impact are substantial. These are the ten advantages most often cited by patients who chose TMS:
- No anesthesia or sedation. The patient is fully awake and oriented throughout each session.
- No IV access. No needles, no infusions, no controlled substances handled.
- Drive yourself. No chaperone or driver required for any session.
- Work the same day. No recovery period. Many patients schedule sessions on lunch breaks.
- No dissociation. Patients with PTSD, dissociation history, or psychotic-spectrum concerns who are excluded from ketamine often qualify for TMS.
- No DEA scheduling burden. No urine screens, no controlled-substance protocols, no Spravato REMS overhead at the clinic.
- No risk of substance-use disorder. TMS has no addiction potential — relevant for patients with active or remitted SUD.
- Insurance pays. Most major commercial plans, Medicare Part B, and Tricare cover TMS for treatment-resistant depression.
- No need to discontinue current antidepressants. Most patients continue their SSRI or SNRI throughout the course.
- Adolescent eligibility. Ages 15 to 21 have FDA-cleared access via NeuroStar (since 2024) and BrainsWay (since 2025).
The corollary disadvantage: TMS demands 30 to 40 visits over 6 to 9 weeks, compared with 6 to 8 visits over 2 to 3 weeks for a typical ketamine induction. TMS is logistically lighter per visit but heavier per course. Patients whose schedules cannot absorb daily visits should consider ketamine therapy or accelerated TMS protocols where available.
Insurance Coverage — Carrier-by-Carrier Reality
TMS is the rare interventional psychiatry treatment where insurance is the default route. Cash-pay is the exception. Coverage criteria vary across the major commercial carriers, and a single difference — UnitedHealthcare’s stricter 3-trial requirement — has meaningful prequalification implications.
| Carrier | Failed Antidepressant Threshold | Coverage Notes |
|---|---|---|
| BCBS | 2 adequate medication trials or evidence-based psychotherapy without sufficient relief | Each Blue plan has its own medical policy; standardized rating-scale documentation required (PHQ-9, HAM-D) |
| Aetna | 2 antidepressant trials or evidence-based psychotherapy | Must use FDA-cleared device in Aetna-certified facility (Clinical Policy Bulletin 0469) |
| Cigna | 2 unsuccessful trials (medication or psychotherapy) | Requires DSM-5 MDD diagnosis from a psychiatrist; participating facility |
| UnitedHealthcare | 3 failed medication trials — most stringent of the major carriers | Requires baseline and follow-up depression scale scores |
| Anthem | 2 to 4 trials depending on plan and state | Pre-auth required; rating-scale documentation required |
| Humana | 2 or more failed trials | Pre-auth required |
| Medicare Part B | 4 failed antidepressant trials from at least 2 different drug classes per most LCDs | Covered nationwide; CMS Article A57072 governs billing |
| Medicaid | Varies by state — significant differences | NY Medicaid added TMS coverage in July 2025; many states still classify TMS as experimental |
| Tricare | Covered for MDD with comparable criteria | Active-duty service members and dependents eligible |
The CPT Codes to Know
Three CPT codes govern TMS billing in 2025 and 2026:
- 90867 — Initial session, includes cortical mapping, motor threshold determination, delivery, and management. Billed once per episode of care.
- 90868 — Subsequent session, delivery and management. The workhorse code, billed for most of the 36 sessions.
- 90869 — Subsequent motor threshold re-determination plus delivery and management. Used when re-mapping is clinically indicated.
When verifying coverage with your insurer, ask specifically about 90867, 90868, and 90869. CMS rules require that these codes never appear together on the same date of service.
What to Do if You Are Denied
About 30 to 50 percent of initial pre-authorization requests are denied. The most common reasons are insufficient documentation of prior medication trials (especially trial duration and dose adequacy), classification as “not medically necessary,” and “experimental” labeling for off-label conditions. The single highest-yield appeal lever is a peer-to-peer review between your clinic’s psychiatrist and the insurer’s medical reviewer; success rates exceed 60 percent when full prior-trial documentation is supplied. Ask your TMS clinic whether they handle peer-to-peer appeals as part of intake.
What TMS Costs — With and Without Insurance
Pricing varies significantly by region and protocol. Major-metro clinics in NYC, San Francisco, Los Angeles, and Boston charge 30 to 50 percent more than midwest and southeast clinics. Cash-pay courses are roughly 25 to 50 percent cheaper than the equivalent insurance-billed amount.
| Scenario | Per Session | Full Course |
|---|---|---|
| Cash-pay national average | $300 to $500 | $7,200 to $15,000 (36 sessions) |
| Cash-pay premium metro (NYC, SF, LA, Boston) | $400 to $600+ | $14,400 to $21,600 |
| With insurance, met deductible (PPO) | $0 to $50 typical | $500 to $1,500 typical |
| With insurance, pre-deductible HDHP | $120 to $250 (negotiated rate) | $3,000 to $6,000 until OOP max hit |
| HMO with mental health rider | $20 to $80 | $500 to $2,500 |
| Medicare Part B (post-deductible) | 20% coinsurance on negotiated rate | Often near-zero with supplemental coverage |
| SAINT cash-pay (outpatient) | — | $30,000 to $36,000 (5 days, 50 sessions) |
| SAINT in hospital outpatient (CMS, 2025+) | — | $19,703 reimbursed to hospital |
Most patients with employer-sponsored insurance hit their out-of-pocket maximum somewhere mid-course — meaning the final 10 to 15 sessions are effectively free under typical ACA-compliant plans. TMS is an HSA- and FSA-eligible expense under IRS Section 213(d) when prescribed for a diagnosed condition, which lets cash-pay patients reduce effective cost. For a deeper dive on cost variance, see our breakdown of TMS therapy costs.
Side Effects and Contraindications
TMS has one of the cleanest safety profiles in interventional psychiatry. The most common side effects are mild and transient.
Common Side Effects (more than 5 percent of patients)
- Scalp discomfort or pain at the stimulation site during sessions — magnetic pulses cause underlying scalp muscles to contract
- Tension-type headache during or after sessions; typically resolves within hours and decreases substantially by week 2 as scalp nerves desensitize
- Facial muscle twitching during stimulation; ends with the session
- Lightheadedness or fatigue post-session
Less Common (1 to 5 percent)
- Vasovagal response (faintness during stimulation)
- Tinnitus or hearing changes if hearing protection is inadequate; ear protection is mandatory
- Tooth or jaw pain from sympathetic nerve stimulation
- Transient mood worsening — the patient-coined “TMS dip”
Rare (less than 1 percent)
- Treatment-emergent hypomania or mania, particularly in undiagnosed bipolar patients; usually resolves with treatment cessation
- Seizure — early-era literature placed risk at approximately 0.1 percent; current real-world data places it closer to 1 in 89,000 sessions
Hard Contraindications — Specific Implants and Conditions
The following are absolute contraindications to TMS treatment due to the risk of magnetic-field interaction with implanted devices or pre-existing neurological conditions:
- Cochlear implants
- Deep brain stimulators (DBS)
- Aneurysm clips, coils, or stents in the brain
- Cranial metal plates or ferromagnetic skull fixation hardware
- Bullet fragments or shrapnel in the head
- Implanted cardiac pacemakers or implantable cardioverter-defibrillators (ICDs) — relative-to-absolute depending on coil distance
- Active seizure disorder or epilepsy — relative-to-absolute
- Recent stroke within 6 to 12 months
- Increased intracranial pressure
Hip and knee replacements, dental fillings, spinal hardware, and other ferromagnetic metal outside the head and neck are not contraindications. Certain medications — bupropion at high doses, clozapine, tramadol, theophylline, and some stimulants — can lower seizure threshold and may require careful titration. Untreated bipolar disorder without mood-stabilizer coverage carries a hypomania-flip risk that warrants pre-treatment review with a psychiatrist.
The “TMS Dip” — What Every Patient Should Know
The “TMS dip” is a patient-coined term — clinics did not invent it, and many do not address it. Some patients experience a temporary worsening of mood or symptom intensification roughly mid-course (commonly between sessions 10 and 20) before clinical improvement begins. The phenomenon resolves with continued treatment in most cases. It is not in itself a sign that TMS is failing.
If you experience the TMS dip, tell your clinician immediately. They may adjust intensity, frequency, or coil position. Importantly, dropping out before session 20 substantially reduces overall response rate — patients who push through the mid-course dip generally have better outcomes than those who quit early. This is one reason continuity matters: TMS works on accumulated cortical effects, not single-session impact.
Pregnancy and Special Populations
TMS is one of the few non-pharmacologic interventions with a growing evidence base for perinatal major depressive disorder. A 2025 Brain and Behavior systematic review and the Clinical TMS Society pregnancy registry — 33 pregnancies with 16 outcome-tracked cases — reported 10 remitted, 6 responded, and no major adverse events. Eighteen-to-sixty-two-month follow-up of children born to treated mothers showed normal cognitive and motor development. Larger multicenter trials remain in progress. The current clinical standard is shared decision-making with maternal-fetal medicine.
For adolescents ages 15 to 21, NeuroStar (March 2024) and BrainsWay Deep TMS (November 2025) hold FDA clearances as adjunct treatment for major depressive disorder. The 12-and-older claim that some clinic content uses is incorrect — adolescent FDA clearance is 15 to 21 only. Real-world evidence from NeuroStar’s 2024 clearance documentation cited 78 percent of treated adolescents achieving clinically meaningful improvement.
Off-Label Uses — Where Evidence Stands
Beyond FDA-cleared conditions, TMS has been studied for several off-label applications. The strength of evidence varies considerably, and patients should know that off-label use generally means cash-pay (insurers typically deny on “experimental” grounds for non-cleared indications).
- Post-traumatic stress disorder (off-label use; not FDA-cleared for this indication) — Moderate-to-strong evidence. A 2025 multisite cohort study of 756 veterans reported 63 to 78 percent response rates and 47 to 49 percent remission across protocols. Stanford’s fMRI-guided personalized PTSD trial published positive results in the American Journal of Psychiatry in 2025.
- Generalized anxiety and social anxiety (off-label use) — Moderate evidence; multi-site trials in progress.
- Bipolar depression (off-label use) — Moderate evidence with hypomania-flip caution.
- Fibromyalgia (off-label use; not FDA-cleared for this indication) — Moderate evidence; randomized trials show roughly 30 percent pain-intensity improvement vs sham, with fatigue reduction.
- Chronic migraine prophylaxis (off-label use) — Moderate evidence. Distinct from the FDA-cleared single-pulse handheld device for acute migraine with aura.
- Addiction (off-label use) — Emerging evidence in cocaine, alcohol, and opioid use disorders. Smoking cessation is the only addiction-related FDA clearance.
- Post-stroke motor recovery (off-label use) — Moderate evidence; primarily academic and research settings.
- Tinnitus, Parkinson’s disease, Alzheimer’s disease (off-label use; investigational) — Limited evidence; some clinics market these aggressively, but insurance will deny on “experimental” grounds.
Is TMS Right for You? — A Decision Framework
Choose TMS if:
- You have insurance and want it to pay. Most major commercial plans, Medicare Part B, and Tricare cover TMS for treatment-resistant depression.
- You have tried at least 2 antidepressants without adequate response. This is also literally the insurance gate for most carriers.
- You can commit to 36 visits over 6 to 9 weeks (or 5 days for SAINT, where available).
- You cannot take significant time off work and do not want a chaperone for sessions.
- You want a non-drug intervention that does not interact with your current SSRIs or SNRIs.
- You have a history of substance use disorder or dissociation that disqualifies you from ketamine therapy.
- You are between ages 15 and 21 with major depressive disorder and seek an FDA-cleared option (NeuroStar or BrainsWay).
Look elsewhere if:
- You have ferromagnetic metal in or near your head — cochlear implants, deep brain stimulators, aneurysm clips, cranial fixation plates, ICDs, embedded shrapnel — all hard contraindications.
- You have an active seizure disorder, recent stroke (within 6 to 12 months), or recent traumatic brain injury.
- You need rapid relief, within days or weeks. TMS takes 2 to 4 weeks to start working; full benefit appears at 6 to 9 weeks. Ketamine, Spravato, or accelerated SAINT are better fits for acute stabilization.
- You cannot commit to 5 sessions per week for 6 to 9 weeks. Drop-out before session 20 substantially reduces response rate.
- You are seeking treatment for a condition without FDA clearance and your insurance will not cover off-label.
How to Find a TMS Clinic
Finding a qualified TMS clinic involves more than proximity. The device a clinic operates determines what conditions it can treat on-label, and the protocols offered determine session length and overall course time. Before booking a consultation, verify these specifics with the clinic:
- Which device do they operate? NeuroStar, BrainsWay Deep TMS, MagVenture, Magstim, or Nexstim. For OCD, only Deep TMS H7 has standalone FDA clearance.
- Which protocols are available? Standard 10 Hz rTMS, iTBS (3-minute protocol), Deep TMS, accelerated TMS, or SAINT. SAINT requires Magnus credentialing.
- Do they handle prior authorization? Ask about peer-to-peer review success rate.
- Are they in-network with your insurer? Verify before scheduling.
- What is the response and remission rate at this clinic? Reputable clinics track outcomes.
- What does maintenance look like after the acute course? Booster sessions, PRN protocols, or combined with antidepressant maintenance.
Search verified TMS clinics in the HealingMaps TMS clinic directory. For patients also considering ketamine therapy or Spravato, the ketamine clinic directory and the Spravato REMS-certified directory list providers by metro and state.
Frequently Asked Questions
How much does TMS cost without insurance?
Cash-pay rates run $300 to $500 per session in most U.S. metros, with premium metros (NYC, San Francisco, Los Angeles, Boston) charging $400 to $600 or more. A full 36-session course costs $7,200 to $15,000 nationally and $14,400 to $21,600 in premium metros. Many independent clinics offer 10 to 25 percent package discounts. Stanford SAINT cash-pay runs $30,000 to $36,000.
Is TMS covered by my insurance?
Most major commercial plans (BCBS, Aetna, Cigna, UnitedHealthcare, Anthem, Humana), Medicare Part B, Tricare, and a growing list of state Medicaid programs cover TMS for treatment-resistant depression. UnitedHealthcare requires 3 failed medication trials; most other carriers require 2. Pre-authorization is mandatory across all major carriers.
What does TMS feel like during a session?
Patients describe a tapping or knocking sensation on the scalp at the stimulation site. The pulses are loud, so hearing protection is mandatory. Some patients feel facial muscle twitching during stimulation; this stops when the session ends. After session 2 to 3, scalp sensitivity decreases substantially.
Can I drive home after TMS?
Yes. Patients drive themselves to and from sessions. There is no anesthesia, no sedation, and no recovery period. Most patients return to work, school, or normal activities immediately afterward. This is the largest patient-experience advantage TMS holds over ketamine therapy and ECT.
What is the “TMS dip”?
Some patients experience temporary mood worsening roughly mid-course (between sessions 10 and 20) before improvement begins. This is patient-coined as the “TMS dip.” It usually resolves with continued treatment. Tell your clinician if it occurs; they may adjust intensity or coil position. Do not drop out — patients who push through generally see better outcomes than those who quit early.
Can I have TMS if I have metal in my head?
Cochlear implants, deep brain stimulators, aneurysm clips, cranial metal plates, embedded shrapnel, and implantable cardioverter-defibrillators are absolute contraindications. Hip and knee replacements, dental fillings, and spinal hardware are not contraindications. Disclose all implants to your clinician during intake.
How does TMS compare to ketamine therapy?
TMS is insurance-covered, non-dissociative, and requires no chaperone or recovery time, but demands 36 visits over 6 to 9 weeks. Ketamine is faster (6 to 8 visits over 2 to 3 weeks for induction) and works in days rather than weeks, but is mostly cash-pay, requires monitored sessions, and produces dissociation. See our detailed TMS-versus-ketamine comparison.
Does TMS work for OCD?
Yes — but only BrainsWay Deep TMS using the H7 coil holds a standalone FDA clearance for OCD (granted via the De Novo pathway in 2018). MagVenture’s Cool D-B80 has an OCD adjunct clearance. If you are seeking TMS for OCD, ask the clinic specifically which device they use and confirm it has the relevant FDA clearance for your condition.
Can teens or adolescents get TMS?
The FDA has cleared TMS for adolescents ages 15 to 21 with major depressive disorder. NeuroStar received clearance in March 2024 (the first adolescent TMS clearance) and BrainsWay Deep TMS in November 2025. There is no current FDA clearance for ages 12 through 14.
What is the success rate of TMS for treatment-resistant depression?
Standard rTMS produces a response rate of 50 to 60 percent and a remission rate of 30 to 40 percent in patients with treatment-resistant depression. Two-thirds of responders maintain benefit at 6 to 12 months. About one-third of responders see depression return within 12 months, with median time-to-relapse of approximately 6 months. Maintenance booster sessions reduce one-year relapse rates from over 32 percent to roughly 16 percent.
Sources and Further Reading
For authoritative medical information about TMS therapy, the National Institute of Mental Health, Cleveland Clinic, and Mayo Clinic maintain comprehensive patient education resources. The FDA’s 510(k) device database documents the clearance history of every TMS device cleared in the United States.
For the original randomized clinical trials supporting key clearances, see the THREE-D trial publication establishing iTBS non-inferiority to standard 10 Hz rTMS, the Stanford SAINT primary publication in PubMed, and the manufacturer pages for NeuroStar, BrainsWay, MagVenture, and Magstim. The Clinical TMS Society maintains professional practice resources at clinicaltmssociety.org.
Last verified: 2026-05-07. This guide is for informational purposes and does not constitute medical advice. Always consult a licensed psychiatrist or healthcare provider before starting any treatment for a mental-health condition.