BPC-157 vs. TB-500: The 2026 Recovery Peptide Comparison
Last reviewed and updated: May 5, 2026 — medically reviewed by Dr. Jonathann Kuo, MD.
2026 Regulatory Update: On April 15, 2026 the FDA removed both BPC-157 and TB-500 from its Category 2 restricted list. Both peptides are scheduled for formal Pharmacy Compounding Advisory Committee review on July 23–24, 2026 — BPC-157 for ulcerative colitis, TB-500 for wound healing. Licensed 503A compounding pharmacies can fill prescriptions from clinicians in most states today. Read our full breakdown of the July 2026 review.
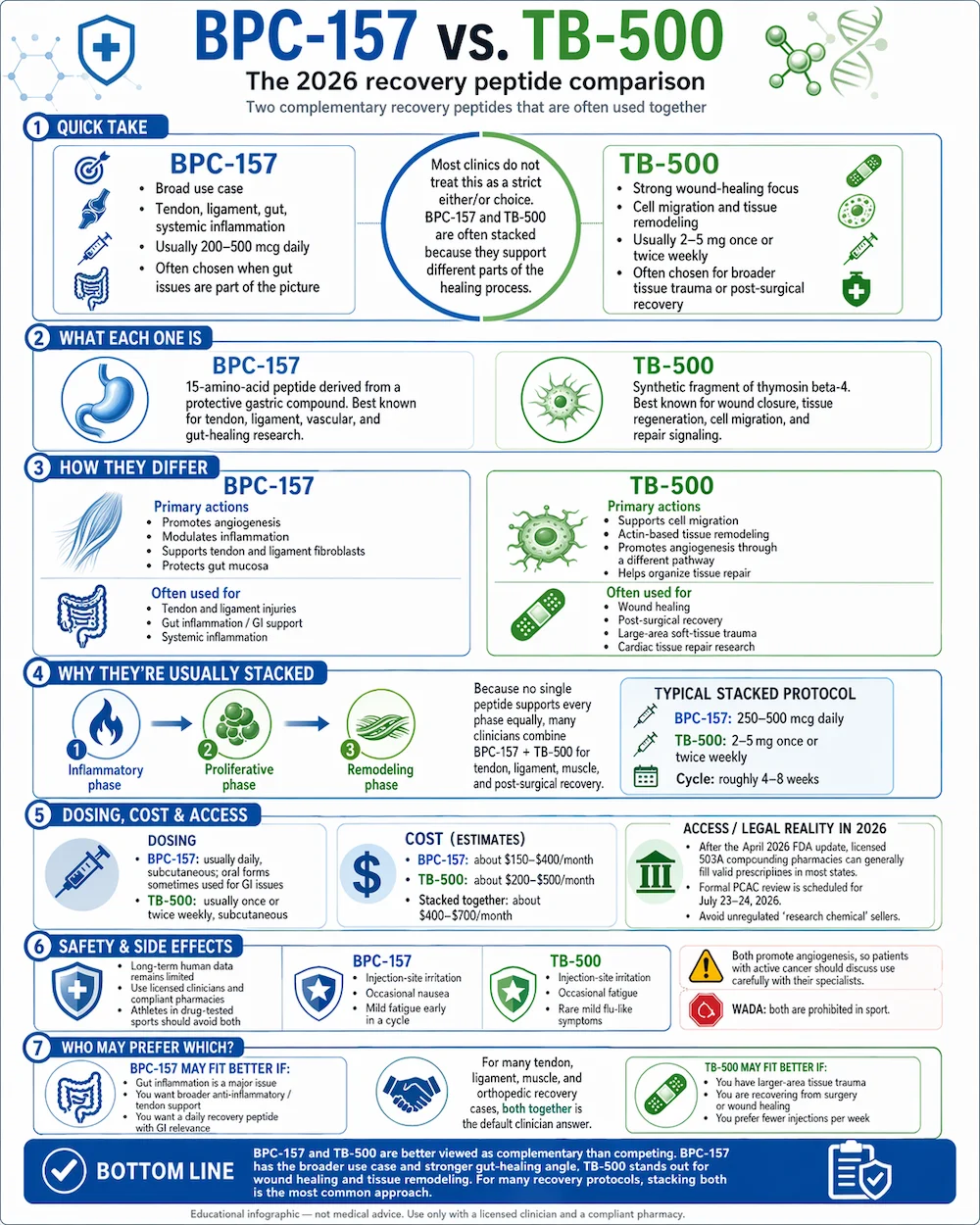
Of all the questions patients ask peptide clinicians, “should I take BPC-157 or TB-500?” is probably the most common. The short answer most clinicians give surprises people: most of them take both. These two peptides solve different parts of the tissue-repair problem, and stacking them is the dominant recovery protocol in peptide clinics today. This guide walks through the real differences, where each shines on its own, and why combining them has become the default.
Looking for a clinic? Find verified BPC-157 and recovery peptide clinics near you.
The Short Answer
BPC-157 has the broader use case — tendons, ligaments, gut healing, systemic inflammation. TB-500 has the stronger evidence for wound healing and cardiac tissue repair. They work through different mechanisms that complement each other, which is why most recovery-peptide clinics prescribe them as a stack rather than asking patients to choose. If you have to pick one, the decision usually comes down to whether your primary issue is soft-tissue injury (either works) or gut inflammation (BPC-157).
What Each Peptide Is
BPC-157 stands for Body Protection Compound-157. It is a 15-amino-acid pentadecapeptide derived from a protein found in human gastric juice. Researchers discovered it in the 1990s while studying how the stomach protects itself from acid damage. The compound extends that protective, regenerative capacity throughout the body — particularly to tendons, ligaments, the vascular system, and the gut lining.
TB-500 is a synthetic fragment of thymosin beta-4 (Tβ4), a 43-amino-acid protein found in nearly every cell of the human body. Tβ4 plays a central role in how cells move, how wounds close, and how tissue regenerates after injury. TB-500 reproduces the active region of that protein for therapeutic use.
How They Differ: Mechanism and Primary Uses
The easiest way to understand the two peptides is to look at what they actually do at the cellular level.
BPC-157 drives angiogenesis and anti-inflammatory signaling. It promotes the formation of new blood vessels at injury sites, modulates nitric oxide production, and upregulates growth-hormone receptors on tendon and ligament fibroblasts. In gut tissue, it protects the mucosal lining from acid and NSAID damage. Its strongest preclinical evidence is in tendon and ligament healing, gastrointestinal ulcer protection, and inflammatory bowel conditions.
TB-500 drives cell migration and actin-based tissue remodeling. It binds G-actin and helps move it where the body needs it for cellular movement during repair. That single interaction has downstream effects across several healing pathways: it supports stem-cell migration to injury sites, promotes angiogenesis (like BPC-157, but through a different mechanism), and dampens inflammatory signaling during the repair phase. Its strongest preclinical evidence is in wound closure, cardiac tissue recovery, and corneal injury repair.
The overlap — tendon and ligament recovery — is why the two peptides are often compared. The non-overlap is why they are usually stacked.
Why They’re Usually Stacked
Tissue healing is a multi-phase process. There’s an initial inflammatory phase, a proliferative phase (new cells and blood vessels form), and a remodeling phase (the tissue reorganizes to something like its original state). No single peptide optimally supports every phase. The case for stacking BPC-157 and TB-500 comes down to this: they work on different phases through different mechanisms, so combining them addresses more of the healing cascade than either alone.
A typical stacked protocol looks like BPC-157 daily (usually 250–500 mcg subcutaneous) plus TB-500 once or twice weekly (usually 2–5 mg subcutaneous) for 4 to 8 weeks. Most clinicians pair the stack with specific rehab work — physical therapy for tendon or ligament injuries, anti-inflammatory diet changes for gut issues, or graded return-to-activity programs for post-surgical patients. Some protocols add a growth-hormone peptide like CJC-1295/Ipamorelin for active adults who want sleep and body-composition benefits alongside recovery.
Dosing and Administration
BPC-157 is most often administered via subcutaneous injection with a small-gauge needle, typically once daily. Some protocols split the daily dose into morning and evening injections. Dosing usually runs 200 to 500 micrograms per day, depending on the condition and patient response. For gastrointestinal conditions specifically, some clinicians use oral capsules, since the peptide acts directly on the gut lining.
TB-500 is also administered via subcutaneous injection, but the schedule is different: one or two injections per week rather than daily. Per-injection doses are larger (usually 2 to 5 milligrams) but the total weekly peptide load is roughly comparable to BPC-157 by cost. Some clinicians favor TB-500 for patients who want fewer injections per week — that’s a real quality-of-life consideration over a 4 to 12 week cycle.
Both peptides are almost always sourced through 503A or 503B licensed compounding pharmacies. Patients should avoid online vendors selling them as “research chemicals” — those products are not tested for human use and have been found in independent testing to vary dramatically in purity and concentration.
Cost and Access
Typical compounded BPC-157 pricing in 2026 runs $150 to $400 per month. Compounded TB-500 runs slightly higher at $200 to $500 per month. Stacked together, patients are usually looking at $400 to $700 per month for both peptides during an active recovery cycle. Almost no insurance covers compounded peptide therapy, so these are cash-pay figures.
The legal access landscape shifted meaningfully in April 2026 when the FDA removed both peptides from its Category 2 restricted list. That opened a clearer pathway for 503A compounding pharmacies to fill prescriptions from licensed clinicians. The July 2026 PCAC review will determine whether either peptide is added to the permanent 503A Bulks List. In the meantime, access is workable but requires both a qualified prescriber and a compliant pharmacy.
Safety and Side Effects
Both peptides have favorable safety profiles in animal studies and in the human case-series data clinicians have accumulated. Side effects are uncommon and typically mild:
- BPC-157: Injection-site irritation, occasional transient nausea, mild fatigue in the first week of a cycle.
- TB-500: Injection-site irritation, occasional fatigue in the first week, rarely mild flu-like symptoms.
Long-term human safety data for both peptides remains limited. Animal data covers relatively short treatment cycles, and extrapolating beyond that requires clinical judgment. Most clinicians cycle patients off the peptides for equivalent periods after a treatment course to allow the body’s signaling to normalize.
Both peptides promote angiogenesis. That is generally desirable for tissue repair, but could theoretically influence tumor environments. Patients with active cancer should discuss peptide therapy carefully with their oncologist. This is not a universal contraindication but it warrants a specialist conversation.
Sports Use and WADA Status
Athletes subject to World Anti-Doping Agency (WADA) testing should not use either peptide during competition. Both are prohibited in sport:
- BPC-157 is classified under WADA category S0 as an “unapproved substance” — banned in and out of competition.
- TB-500 is classified under WADA category S2 as a “peptide hormone, growth factor, related substance and mimetic” — also banned in and out of competition.
Detection windows vary by test and protocol. Athletes in drug-tested sports should consult a sports medicine physician familiar with WADA rules before considering either compound.
Who Should Consider Which?
If you’re deciding rather than stacking, the rough guidelines most clinicians use are:
- BPC-157 alone — patients whose primary issue is gut inflammation, inflammatory bowel conditions, NSAID-induced stomach damage, or broader systemic inflammation where the gut is the focal point.
- TB-500 alone — patients with large-area wound healing needs, post-surgical patients with significant tissue trauma, or those focused on cardiac tissue recovery after injury.
- Both together — the default recommendation for tendon, ligament, and muscle injuries, post-orthopedic-surgery recovery, chronic soft-tissue problems that haven’t responded to standard care, and general athletic recovery programs.
Frequently Asked Questions
Which works faster, BPC-157 or TB-500?
Both peptides typically produce first noticeable effects — reduced pain, improved range of motion, better sleep — within 2 to 3 weeks of starting a cycle. Full tissue-level effects generally take the length of the cycle, 4 to 12 weeks. Neither is meaningfully faster than the other in the early weeks.
Can I take them orally?
Oral BPC-157 capsules are available through some compounding pharmacies and are considered reasonable for gastrointestinal conditions, since the peptide acts directly on the gut lining. For systemic tissue repair (tendons, ligaments, joints), subcutaneous injection is more reliable for both peptides. Oral TB-500 is not commonly used.
Do I need to refrigerate them?
Yes. Both peptides are typically supplied as lyophilized (freeze-dried) powder that the patient or pharmacy reconstitutes with bacteriostatic water. The reconstituted solution should be stored refrigerated and used within the stability window specified by the compounding pharmacy — usually 30 days.
What other peptides are commonly stacked with BPC-157 or TB-500?
For active adults focused on recovery plus body composition, clinicians often add a growth-hormone peptide like CJC-1295 / Ipamorelin. For patients with significant gut inflammation alongside joint issues, clinicians may layer KPV. For post-illness recovery, some protocols include Thymosin Alpha-1 for immune support.
Is it safe to cycle off after a recovery protocol?
Yes — and most clinicians recommend it. A typical approach is 4 to 12 weeks on, then an off-cycle of equivalent length. This gives the body’s endogenous repair signaling time to normalize between courses. Patients with chronic conditions may run more frequent cycles under clinician supervision.
What should I ask a clinic before starting?
Four questions to ask: (1) Is the compounding pharmacy 503A or 503B licensed? (2) Does the pharmacy provide third-party testing for purity, sterility and concentration? (3) Will the clinic order baseline labs before prescribing? (4) What is the follow-up protocol to monitor response and side effects? A reputable recovery-peptide clinic will answer all four clearly.
The Bottom Line
BPC-157 and TB-500 are complementary rather than competing peptides. BPC-157 has the broader use case and the stronger gut-healing evidence. TB-500 has the stronger evidence for wound closure and cardiac tissue. Most recovery-peptide clinics prescribe them as a stack because the two compounds address different phases of the healing cascade. The April 2026 FDA reclassification has made legitimate access — through licensed clinicians and compliant compounding pharmacies — more straightforward than it has been in years. For patients dealing with stubborn soft-tissue injuries, post-surgical recovery, or chronic inflammation, either peptide is worth discussing with a qualified provider. Both is usually the answer they give back.
Ready to find a clinic? Browse our directory of verified BPC-157 and recovery peptide clinics to find a licensed provider near you.



