Tirzepatide vs. Retatrutide: The 2026 Next-Generation GLP-1 Comparison
Last reviewed and updated: May 5, 2026 — medically reviewed by Dr. Jonathann Kuo, MD.
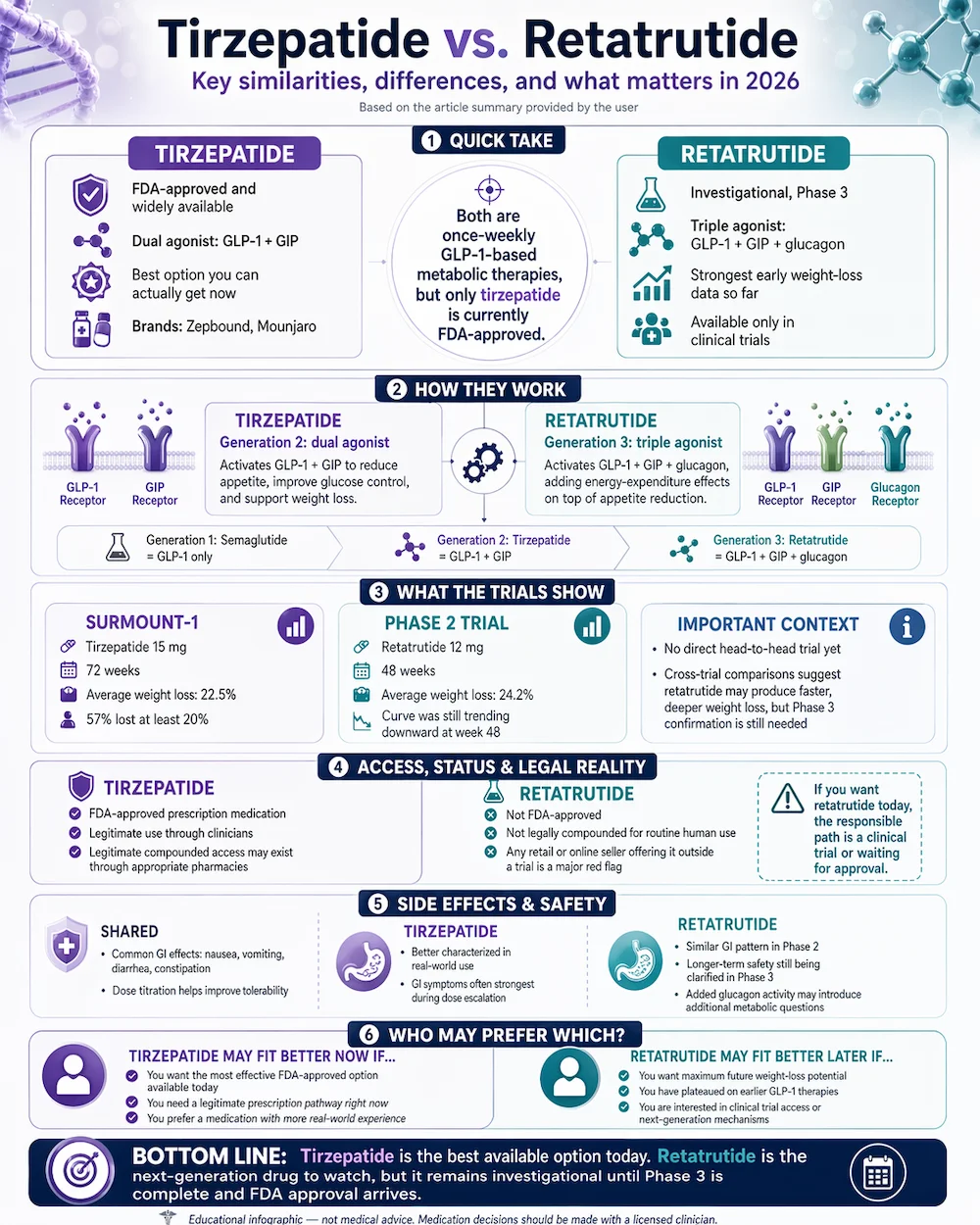
2026 Status: Tirzepatide (Zepbound, Mounjaro) is FDA-approved and widely available. Retatrutide — the next-generation triple-agonist GLP-1 — is in Phase 3 trials. Early results show the most potent weight loss of any GLP-1-class drug studied to date, but it’s not yet legally available outside of clinical trials.
The GLP-1 weight-loss category moves fast. Semaglutide led the market until tirzepatide surpassed it. Tirzepatide currently leads — but retatrutide has already shown numbers in trials that exceed tirzepatide by meaningful margins. If you’re a patient trying to understand the landscape, here’s where things stand in 2026.
Looking for a clinic? Find verified GLP-1 weight-loss clinics near you.
The Short Answer
Tirzepatide is the best option you can actually get right now. Retatrutide has shown higher weight loss in Phase 2 trials and is expected to become the most effective weight-loss drug on the market once approved, but as of April 2026 it remains an investigational compound available only through clinical trials. Anyone selling retatrutide outside of a trial is selling it illegally — it cannot be legally compounded by 503A pharmacies because it’s not FDA-approved.
How They Work — The Mechanism Upgrade
The GLP-1 class has evolved through three mechanism generations:
- Generation 1 (single agonist): Semaglutide binds one incretin receptor, GLP-1. Produces about 15% body weight loss on average.
- Generation 2 (dual agonist): Tirzepatide binds two incretin receptors, GLP-1 and GIP. Produces about 22% body weight loss on average.
- Generation 3 (triple agonist): Retatrutide binds three receptors — GLP-1, GIP, and glucagon. The added glucagon agonism increases energy expenditure in addition to reducing appetite. Produces about 24% body weight loss on average at 48 weeks in Phase 2 trials, with higher doses still being evaluated.
Each generation builds on the previous mechanism. Tirzepatide didn’t replace semaglutide’s GLP-1 agonism — it added GIP on top. Retatrutide adds glucagon on top of the dual mechanism. Whether each generation’s benefits justify the additional side-effect burden is the key clinical question.
What the Trials Actually Showed
Tirzepatide (SURMOUNT-1, NEJM 2022): 2,539 adults with obesity randomized to 5 mg, 10 mg, 15 mg tirzepatide or placebo for 72 weeks. At the 15 mg dose, participants lost 22.5% of body weight on average. Nine of ten patients at the highest dose lost at least 5% of body weight; 57% lost at least 20%. These results made tirzepatide the most effective weight-loss medication ever FDA-approved at that point.
Retatrutide (Jastreboff et al., NEJM 2023): 338 adults with obesity randomized to retatrutide at multiple doses (1, 4, 8, 12 mg) or placebo for 48 weeks. At 12 mg, participants lost 24.2% of body weight on average — and the curve was still trending downward at week 48, suggesting they might have lost more with continued use. Phase 3 trials are testing longer durations and higher doses.
A direct comparison in a single trial doesn’t exist yet. But looking at the data across studies, retatrutide at 12 mg achieves in 48 weeks roughly what tirzepatide at 15 mg achieves in 72 weeks — potentially offering faster, deeper weight loss. Caution: Phase 2 data doesn’t always hold up in Phase 3, side effect profiles can look different at scale, and the added glucagon mechanism may create new considerations.
Regulatory and Legal Reality
This is the part patients need to be careful about. Tirzepatide is FDA-approved. Zepbound, Mounjaro and legitimate compounded versions through 503A pharmacies are legal with a prescription. Retatrutide is not FDA-approved. It cannot be legally compounded by pharmacies for human use. Any vendor selling retatrutide outside of a clinical trial is selling an unapproved drug — the quality, purity, and safety are entirely unverified.
Internet vendors marketing retatrutide as “for research only” or similar language are selling unregulated material that has been found in independent testing to vary dramatically in purity and concentration. This is the same problem the peptide field has had with BPC-157, TB-500 and other non-approved compounds — except retatrutide hasn’t even completed Phase 3, so its safety profile is less well understood than the research peptides that have been studied for decades.
The responsible path if you want retatrutide: enroll in a clinical trial (check ClinicalTrials.gov) or wait for FDA approval, which analysts currently expect in the 2026–2027 window.
Side Effects
Both drugs produce GI side effects as their most common adverse events — nausea, vomiting, diarrhea, constipation. This is the class effect of GLP-1 activation and dose titration makes it manageable in most patients. Tirzepatide’s profile is well characterized across three-plus years of real-world use; GI effects are most intense during dose escalation and usually subside.
Retatrutide’s Phase 2 data showed a similar GI pattern. The added glucagon agonism theoretically raises questions about effects on glucose regulation (especially in patients with diabetes) and cardiovascular parameters. Phase 3 data will clarify these questions. Until then, drawing firm conclusions about retatrutide’s long-term safety would be premature.
Who Should Consider Which?
- Tirzepatide today: Patients seeking the most effective FDA-approved weight-loss option available through legitimate channels. Appropriate for most patients with obesity or type 2 diabetes, with clinician oversight.
- Retatrutide eventually: Patients who have tried other GLP-1 medications and plateaued, patients seeking maximum weight loss, patients interested in the triple-agonist mechanism for specific metabolic reasons. Currently requires trial enrollment.
- Semaglutide still: Patients prioritizing lower cost, longer real-world safety track record, or insurance coverage that favors Wegovy/Ozempic. See our semaglutide vs tirzepatide comparison for that detailed choice.
HealingMaps may earn a commission when readers sign up through ShedRx. This does not affect our editorial coverage or your price. ShedRx’s “lose 10% of your weight or full refund” promise is a contractual refund policy — full money back if you do not lose at least 10% of your body weight on the program.
Frequently Asked Questions
When will retatrutide be FDA-approved?
Eli Lilly’s Phase 3 program (TRIUMPH) is running through 2026 and early 2027. Based on similar drug development timelines, FDA approval is most often projected in the 2026–2027 window, but regulatory timelines can shift.
Is retatrutide the same as tirzepatide plus something?
Mechanistically similar but not a combination product. Retatrutide is a single molecule designed to activate all three receptors (GLP-1, GIP, glucagon) in balanced ratios. Tirzepatide is a single molecule that activates GLP-1 and GIP only. Both are synthetic peptides engineered for their specific receptor profiles.
Will retatrutide replace tirzepatide once approved?
Probably not entirely. Tirzepatide will likely remain first-line for many patients due to cost and established track record. Retatrutide is likely to become the option for patients who need maximum weight loss or who haven’t responded adequately to dual agonists. The GLP-1 market will become segmented by mechanism generation and price.
Can I get compounded retatrutide now?
No. The FDA compounding rules require that a drug be approved before it can be compounded. Retatrutide is not FDA-approved. Any vendor selling compounded retatrutide is doing so illegally and the product quality is unverifiable.
Is it worth waiting for retatrutide?
Generally no — patients who would benefit from weight loss today shouldn’t wait 1–2 years for a theoretically better option. Tirzepatide is the best available FDA-approved drug. Retatrutide is likely to be incrementally better when approved, but the health benefits of losing weight today typically outweigh the marginal improvement of waiting.
The Bottom Line
Tirzepatide is today’s best-available weight-loss option — FDA-approved, widely prescribed, well-characterized, and legitimately compoundable for patients who need lower-cost access. Retatrutide is the next generation. Phase 2 data shows it outperforming tirzepatide in side-by-side comparison of their respective trials, and Phase 3 results over the next 12–18 months will clarify whether that advantage holds. For patients making a decision today, the answer is tirzepatide with a qualified clinician. For patients following the category’s evolution, retatrutide is the drug to watch through 2026–2027.
Ready to start with what’s available today? Browse our directory of verified GLP-1 and peptide therapy clinics to find a licensed provider.



